8 years
Risk of Osteosarcoma with Anabolic Therapy
Unfolding the Truth
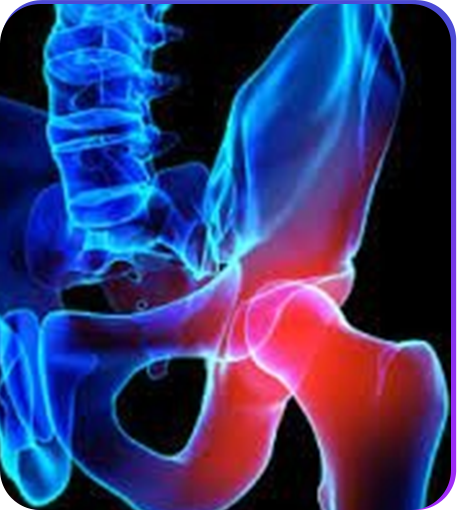



Burden of Osteoporosis
Global Prevalence

> 200 million individuals suffer from osteoporosis globally1
Indian Prevalence





1 out of 5 Indians suffer from osteoporosis2

References:
- Sozen T et al. European journal of rheumatology.2017;4(1):46-56
- Available at: https://ijoro.org/index.php/ijoro/article/view/1881. Accessed on 17th June,2021 at 5:45pm IST



Need of Anabolic Therapy in Osteoporosis
T-score
Anabolic therapy is indicated for the patients who are at a very high-risk1
(Multiple vertebral fractures in addition to a bone mineral desity T-score of > -2.5)
References:
- https://www.endocrineweb.com/professional/meetings/anabolic-agents-superior-first-line-therapy-highest-risk-patients
- Accessed on 25th June, 2021 at 1:34pm IST



Approved Anabolic therapies1


Teriparatide
Abaloparatide
Romosozumab (under reviewby the USFDA)
Impact of Anabolic therapy in Osteoporosis2


Daily PTH injections or PTH1-34
↑Serum PTH concentration
Bone formation Bone resorption to minor extent
↑Bone mass ↑Bone microarchitecture ↑Mechanical strength

USFDA: United States Food and Drug Administration | PTH Parathyroid Hormone
References:
- Haas AV et al. Journal of the Endocrine Society.2018;2(8):922-932
- Russow G et al. International journal of molecular sciences.2019;20(1):83-100



Contraindications of Teriparatide and Abaloparatide
(Food and drug Administration and European Medicines Agency study)1
A history of bone cancer (primary or metastatic)
Risk of Osteosarcoma
Children and young adults with open epiphysis
Reference:
- Vahle JL et al. Toxicologic pathology.2002;30:312-321



Clinical Evidence: Risk vs No Risk of Osteosarcoma with Teriparatide(1/3)
Outdated evidence on use of anabolic therapy1
An animal study conducted in 2012 concluded that: ~26% incidence of osteosarcoma was associated with anabolic therapy administered for a period of > 18-24 months
Reference:
- Capriani C et al. Journal of bone and mineral research.2012;27(12):2419-2428



Clinical Evidence: Risk vs No Risk of Osteosarcoma with Teriparatide(2/3)
Latest Post-marketing surveillance of osteosarcoma in patients receiving Teriparatide
10 years
15 years
8 years
10 years
15 years
References:
- Gilsenan A el al. Osteoporosis International.2018;29(10):2335-2343.
- Gilsenan A el al. 2021;32(4):645-651.
- Miller PD el al. Current Medical Research and Opinion.2020;36(11):1861-1872.



Clinical Evidence: Risk vs No Risk of Osteosarcoma with Teriparatide(3/3)
| Studies | Type of study | Drugs with dosage | Study population / Sample size | Follow up period | Incidence of Osteosarcoma |
|---|---|---|---|---|---|
| Gilsenan1 | Prospective study | Teriparatide | 2.4 lakhs | 8 years | No incidence |
| Gilsenan2 | Prospective study | Teriparatide | 75 thousands | 10 years | No incidence |
| Gilsenan3 | Population based comparative-cohort study | Teriparatide | 1.5 lakhs; >= 65 years | 8 years | 0.3.2 times incidence |
| Miller4 | Post marketing surveillance | Abaloparatide | 2 years | No incidence | |
| Gilsenan5 | Post marketing surveillance | Teriparatide | >=40years | 15 years | No difference in incidence |
| Osteosarcoma surveillance safety studies6 | Teriparatide |
3.7 lakhs 1.5 lakhs |
7 years | Three and zero osteosarcoma |
References:
- Gilsenan A et al. Osteoporosis International.2018;29(10):2335-2343.
- Gilsenan A et al. 2021;32(4):645-651.
- Gilsenan A et al. Pharmacoepidemiol Drug Safety.2020;29:1616-1626.
- Miller PD et al. Current Medical Reasearch.2021;36(2):244-251
- Gilsenan A et al. Journal of Bone and Mineral Reasearch.2021;36(2):244-251.
- Available at https://pi.lilly.com/us/forteo-pi.pdf Accessed on 17th June,2021 at 5:45pm IST



Dosage Recommendations of Anabolic agents
Use of Teriparatide for > 2 years should only be considered if a patient remains at or has returned to having a high risk for fracture2
Abaloparatide*
- 80-µg daily subcutaneous injection
- Periumbilical region
- Discontinued after 24 months1
*Not available in India

Removal of Black Box Warning for Osteosarcoma1
References:
- Hass AV et al. Journal of the Endocrine Society.2018;2(8):922-932
- Available at https://pi.lilly.com/us/forteo-pi.pdf. Accessed on 17th June,2021 at 5:45pm IST.



Conclusion
- Anabolic agents are highly effective at increasing bone mineral density and reducing incidence of both vertrbral and nonvertebral fractures
- Anabolic therapy resulting in osteosarcoma continues to be a theoretical issue, however, the clinical experience with both forms of PTH has not substantiated such concerns
PTH: Parathyroid hormone
Reference:
Capriani C et al. Journal of bone and mineral research.2012;27(12):2419-2428



Disclaimer


